pH Modifiers
pH Modifiers
Acid waters are, almost by definition, poor in calcium and often generally low in other ions, but some very soft waters may have a neutral pH (Hynes 1970). It is difficult to separate the effects of high concentrations of hydrogen ions from low base content, and many studies suggest that acidity may never be the major factor controlling the presence or absence of particular plants and animals. Nevertheless, some researchers have demonstrated a good correlation between pH levels and plant distribution (Sjörs 1950; Jeglum 1971). Jeglum (1971) showed that plants can be used to predict the pH of moist peat.
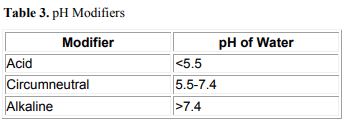
There seems to be little doubt that, where a peat layer isolates plant roots from the underlying mineral substrate, the scarcity of minerals in the root zone strongly influences the types of plants that occupy the site. For this reason, many authors subdivide freshwater, organic wetlands into mineral-rich and mineral-poor categories (Sjörs 1950; Heinselman 1970; Jeglum 1971; Moore and Bellamy 1974). We have instituted pH Modifiers for freshwater wetlands (Table 3) because pH has been widely used to indicate the difference between mineral-rich and mineral-poor sites, and because it is relatively easy to determine. The ranges presented here are similar to those of Jeglum (1971), except that the upper limit of the circumneutral level (Jeglum’s mesotrophic) was raised to bring it into agreement with usage of the term in the U.S. The ranges given apply to the pH of water. They were converted from Jeglum’s moist-peat equivalents by adding 0.5 pH units.
Created with the Personal Edition of HelpNDoc: Easy EBook and documentation generator